Lewis structure of boron trifluoride: Each pair of dots represents a pair of electrons. 
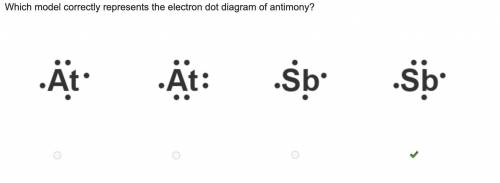
The Lewis Structure uses Hund's rule when placing the dots around the element. Tin? You can follow their steps in the video explanation above. Get a better grade with hundreds of hours of expert tutoring videos for your textbook, By registering, I agree to the Terms of Service and Privacy Policy, Example: Draw the electron-dot symbol for each of the following elements. We subtract 10 electrons to account for Thank you!Status: Resolved. How much does does a 100 dollar roblox gift card get you in robhx? distribute. seesaw Based on our data, we think this question is relevant for Professor Labrake's class at TEXAS. Li Be B C N O F Ne, Concept: Rules for Drawing Molecular Compounds, For SbH3: Antimony (Sb) → EN = 1.9 Hydrogen (H) → EN = 2.2. Follow step-by-step procedure for drawing Lewis structures. A dot diagram (also called an Electron Dot Diagram, and a Lewis Structure) is a way to show the valence electrons that surround an element. We subtract 10 electrons to account for the five bonds in the skeleton leaving us with 30 electrons to distribute. A Lewis dot structure for SeO3 is drawn with an Se in the center, with two lines connecting it to two Os and one double line connecting it to an O. SbF 3 is prepared by treating antimony trioxide with hydrogen fluoride: Sb 2 O 3 + 6 HF → 2 SbF 3 + 3 H 2 O. Antimony is a chemical element with the symbol Sb (from Latin: stibium) and atomic number 51. Antimony trichloride is an inorganic chloride salt with formula SbCl3. SbF3 is also called Antimony trifluoride.For a complete tutorial on drawing Lewis Structures, see my video: more practice, see learn to find the valence electrons: more chemistry help at done in InkScape. Pagkakaiba ng pagsulat ng ulat at sulating pananaliksik? Note: Make sure to not pair them up( meaning put two dots beside each other) unless you have to. Given descriptions, diagrams, and chemical symbols of any element, students will show the arrangement of electrons in the outer-most energy levels by using Lewis valence electron dot structures. Our tutors have indicated that to solve this problem you will need to apply the Lewis Dot Structure concept. Germanium? What is the rising action of faith love and dr lazaro? Antimony (Sb) can be an exception to the octet rule, accepting up to twelve electrons. Our expert Chemistry tutor, Dasha took undefined 38 seconds to solve this problem. A step-by-step explanation of how to draw the SbF3 Lewis Dot Structure. It is characterized as a toxic, colorless gas that is heavier than air with a disagreeable odor, and exposure occurs by inhalation, ingestion, or contact. Once we know how many valence electrons there are in SbF3 we can distribute them around the central atom with the goal of filling the outer shells of each atom.In the Lewis structure of SbF3 structure there are a total of 26 valence electrons. What are some common mistakes students make when drawing Lewis structures? A bond can be drawn as a line between two atoms, which also indicates two electrons. The demand for camping trailers occurs between January and June (mostly in April and May.Selenium trioxide | SeO3 - PubChemSelenium trioxide | SeO3 - PubChem. (Only for elements with 1-4 valence electrons do you have to follow the rule when using the structure. We offer services in the Hunter, Newcastle and Lake Macquarie areas of NSW, in and around these locations: Branxton | Cessnock | Lake Macquarie | Maitland | Morriset | Newcastle | Pokolbin | Port Stephens | Raymond Terrace | Rutherford | The Entrance | TuggerahĬopyright © 2020 Multiply Media, LLC.We offer services on the Mid-North Coast of NSW, in these areas: Bellingen | Coffs Harbour | Coramba | Dorrigo | Grafton | Kempsey | Nambucca Heads | Macksville | Red Rock | Sawtell | Toormina | Urunga | Woolgoolga | Wooli.Be a Community Support Worker at Subee Newlake. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
